What should patients do if they have this medicine at home or are equipped with it in case of need?
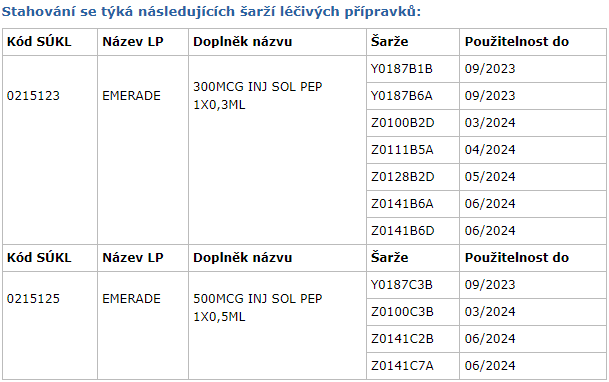
Check the EMERADE batch number found on the medicine packaging.
If you have a batch at home affected by a quality defect, contact your doctor and ask him/her to prescribe a replacement medicine, alternatives are prefilled pens EPIPEN, 300 MCG INJ SOL PEP 1X0.3ML or EPIPEN, 300MCG INJ SOL PEP 2X0.3ML.
The batch affected by the quality defect can be returned without presentation of a receipt at any pharmacy, but preferably the one where the product was dispensed, until 9 August 2023. In order not to endanger the health of patients, the return of the EMERADE pre-filled pen to the pharmacy should only take place after the receipt of the alternative adrenaline pen.
The recall applies to both loaded and unused packs of the product. The recall does not apply to autoinjectors that have already been activated.
If you have any concerns or questions regarding this product, you may contact PharmaSwiss Czech Republic s.r.o., tel: 234 719 600 or e-mail: czech.info@bauschhealth.com.
Can a quality defect endanger the health of patients?
As EMERADE is a life-saving medicinal product, failure to activate the pre-filled pen is assessed as life-threatening. Therefore, the marketing authorisation holder, in cooperation with the CMA, has decided to carry out a recall down to the patient level. For patients who have already used the medicinal product and it has been successfully administered, the product does not pose any risk, the reason for the recall being possible problems with the activation of the pen.
When is a patient entitled to a refund?
The patient is entitled to a refund of the EMERADE, 300MCG INJ SOL PEP, 1X0.3ML supplementary payment of CZK 92 without the need to submit an accounting document by the pharmacy, which will then be reimbursed by the marketing authorisation holder.
Reimbursement for the recalled medicinal product
Replacement with another batch of EMERADE prefilled pen is not currently possible. To avoid compromising patient safety, the return of the EMERADE pre-filled pen to the pharmacy should only take place after the receipt of the alternative adrenaline pen, EPIPEN. The patient should be instructed on the use of the new pen.