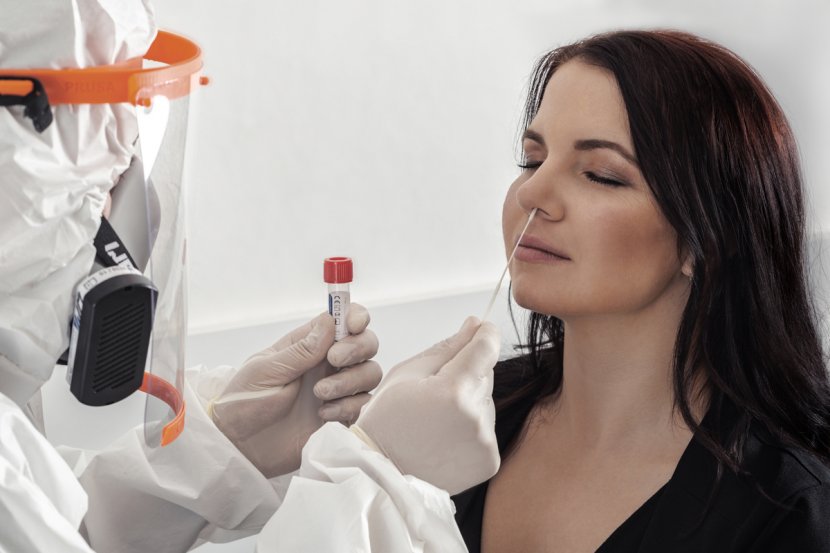
The study was carried out on a sample of 500 patients in collaboration with the 3rd Medical Faculty of Charles University and the company SPADIA LAB, a.s. The data on the reliability of antigen tests declared by the manufacturers proved to be unattainable in clinical practice for many products on the market. The main aim of the study was to independently verify the reliability of specific antigen tests.
"Rapid and affordable testing has the potential to become one of the cornerstones of the fight against the Covid-19 pandemic. We were looking for a reliable method to help our hospitals in clinical decision making. In fact, we wait too long for PCR results and have limited capacity, which severely limits our routine operations. We were disappointed with the results of some of the antigen tests, but in the end we seem to have found a test that is applicable to our practice." said František Vlček, Director of Processes and Quality at AKESO Holding, adding for context, "PCR testing had and still has an irreplaceable role in the Covid-19 epidemic. However, it has its limitations in terms of speed and availability. Antigen testing, of course, only on the condition that it demonstrates a sufficient level of reliability, can fundamentally reverse the balance of power in the fight against the Covid-19 pandemic, and not only in the operation of healthcare facilities. Rapid, accessible and inexpensive testing has proven to be a very effective alternative to blanket bans and restrictions in many foreign countries. In other words, we can either "proscribe" or "protest" the crisis. The latter alternative certainly has less impact on the economy and, more importantly, on our lives."
About the AKESO holding antigen test study
Method
A total of 500 people presenting for RT-PCR testing for SARS-CoV-2 were enrolled in the study at two collection sites operated by the Hořovice Hospital in Hořovice and in Beroun.
Two samples were taken from the study subjects, one was sent for RT-PCR analysis at the SPADIA LAB and the other was analysed on site using the Wantai SARS-CoV-2 Rapid Antigen Test (FIA), a product of Beijing Wantai Biological Pharmacy Enterprise Co., Ltd.
Information on their Covid-19 disease symptoms, duration, comorbidities, and other information was collected from subjects enrolled in the study and will be further statistically evaluated in detail.
Positivity/negativity was evaluated for PCR testing, and the threshold cycle of positivity (Ct) for positive results.
Preliminary results
The validated antigen test for SARS-CoV-2 shows high sensitivity in the general population (91.04 %) and especially in persons with a viral load corresponding to Ct≤35 in RT-PCR (95.45 %). The specificity value is also satisfactory (93.92 %), whereas manufacturers often declare 100 % for other products on the market. The occurrence of false-positive results can be partly explained by the study methodology, where separate sampling was performed for each of the two methods compared, which in several cases in patients with borderline detectable viral load (Ct 35-41) resulted in PCR positivity above Ct 38, which is already considered a negative result according to the methodology of the collaborating laboratory.
The above mentioned reliability parameters of the evaluated test argue for its applicability in routine operation of a healthcare facility or for targeted testing of larger groups of people (education, industry, tourism, etc.).
In the context of the results of other comparative studies already carried out (e.g. by the Institute of Medical Microbiology, 2nd Faculty of Medicine, Charles University and Motol University Hospital), it can be assumed that the quality of antigen tests currently available on the market often differs significantly from the data declared by the manufacturer.
Following the results of this and other studies, it would be advisable to develop, if possible, an objective methodology for assessing the reliability of antigen tests when they are introduced to the market in the Czech Republic.
These are preliminary results. Detailed statistical analysis of the cohort is still in progress and the final results may vary slightly.